With over thirty years of experience manufacturing custom plastic extrusions and clear packaging tubes, Petro Packaging Co., Inc. serves a wide range of medical-related manufacturers across the United States. Our extrusion solutions are made from FDA-approved materials and are well-suited for non-sterile medical packaging and medical device components. Many companies in the medical field rely on Petro Packaging Co., Inc. for high-quality, durable packaging tubes and extrusions to safeguard valuable components during storage and transport. While our manufacturing does not take place in a clean room environment, we have implemented numerous environmental controls in our manufacturing facilities and our materials meet FDA standards for medical and food contact applications.
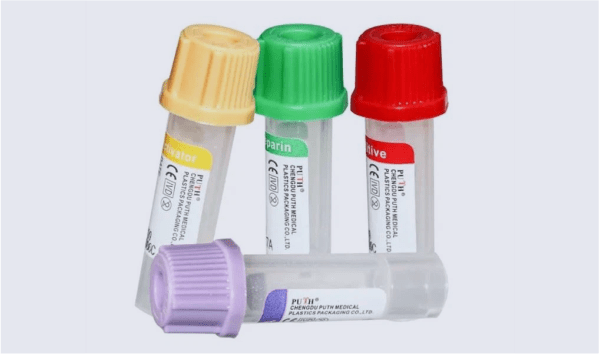
Precision Plastic Medical Packaging & Tubing Solutions
FDA Approved Materials
We specialize in extruding PETG Copolymer (Polyethylene Terephthalate Glycol) and Tenite™ CAP (Cellulose Acetate Propionate) resins – both FDA-approved – for our clear plastic packaging tubes. These materials offer excellent clarity, strength, and chemical resistance. Paired with removable caps or custom end closures, our tubes create a crush-resistant, see-through solution that protects sensitive items from physical damage.
Although we specialize in these materials, we are not limited to them. Contact us regarding custom extrusions with alternate material requirements!
Packaging Tubes and Containers
Our plastic packaging tubes are widely utilized to provide reliable protection for a variety of delicate items, including catheter components, micropipettes, and capillary tubing. In addition to our standard offerings, we manufacture several variations of these tubes to meet different needs. These include threaded tubes that feature a tight fitting metal cap for added security and shaped tubes that offer roll protection.
For customers with specialized packaging requirements, our experienced engineering team is available to work closely with you. We can develop customized solutions designed to accommodate non-standard dimensions, unusual shapes, or specific functional criteria, ensuring that your products are properly protected and packaged according to your exact specifications.
Protective Sleeves & Extruded Components
In addition to tubing, Petro Packaging Co., Inc. also manufactures custom plastic extrusions for protections and structural components used in medical devices. These parts can serve as protective housings, spacers, insulators, or support elements in a wide variety of equipment and instruments. Whether you’re looking for a simple extruded sleeve or a more complex shape, we can work with your design team to deliver parts that meet your specifications using FDA-approved materials.
Our focus is on precision, durability, and versatility—ideal for device manufacturers who need reliable, non-sterile components for assembly or packaging.